Castagna A. AIDS. 2014 Sep 24;28(15):2269-79 ; Galli L. J Antimicrob Chemother. 2016 Jun;71(6):1637-42
Type of ARV Trial
Switch studies in virologically suppressed patients
» Switch to PI/r monotherapy
» ATV/r mono vs ATV/r + 2 NRTI
Switch studies in virologically suppressed patients
» Switch to PI/r monotherapy
» ATV/r mono vs ATV/r + 2 NRTI
Drugs
PI/r mono, ATV/r, 2 NRTI
PI/r mono, ATV/r, 2 NRTI
- ATV/r monotherapy treatment simplification showed lower virological efficacy in comparison with maintaining triple therapy with ATV/r
+ 2 NRTI
- Inferiority more pronounced in those with
- Pre-ART HIV RNA > 100 000 c/mL
- Nadir CD4 < 350/mm3
- HCV co-infection
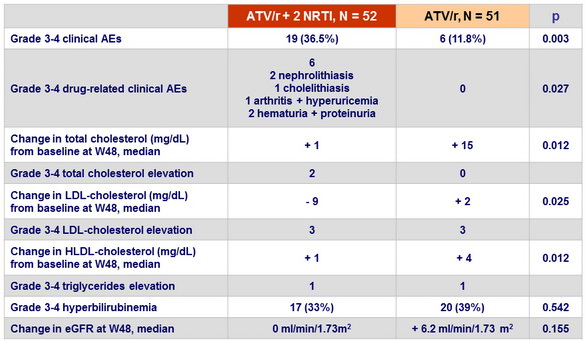
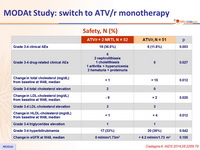
- No benefits of ATV/r monotherapy on lipids or renal function
- Inferiority more pronounced in those with
- NRTIs re-intensification was effective in all the individuals
- The study was terminated early due to recommendation of IDMC
- W96 results
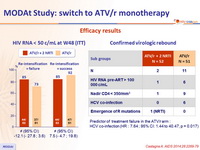
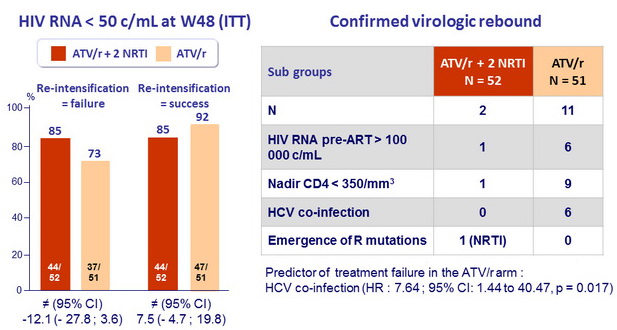
- HIV RNA < 50 c/mL
- ATV/r monotherapy = 64% vs ATV/r + 2 NRTI = 63%
- Difference (95% CI): 1.3% (-17.5 to 20.1%) => monotherapy not non inferior
- ATV/r monotherapy arm
- Median HIV RNA at viral rebound (N=14): 136 (72–376) copies/mL;
- No PI- or NRTI-associated resistance mutations were observed and all patients re-suppressed after reintensification
- Drug-related adverse events leading to discontinuation: 3 (6%) in ATV/r monotherapy arm vs 11 (21.5%) in the triple-therapy arm (p = 0.041)
- The 96 week adjusted mean percentage change in total proximal femur BMD was +1.16% and -1.64% in the ATV/r monotherapy arm and the triple-therapy arm, respectively (p = 0.012)
Design :
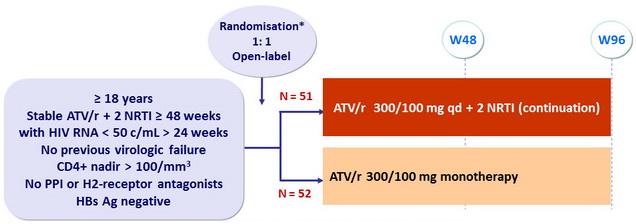
* Randomisation was stratified on HIV RNA (≤ or > 100,000 c/mL) prior to ART start
Objective
- Primary Endpoint : proportion with treatment success at W48
- Treatment failure : treatment discontinuation for any cause or confirmed virologic rebound (first of 2 consecutive HIV RNA > 50 c/mL within 2 weeks)
- Non -inferiority of ATV/ r (ITT analysis ) ; lower limit of the 2-sided 95 % CI for the difference = -10% ; sample size = 342 (171 x 2), 80% power
- Interim analysis by IDMC in June 2013 : recommendation to stop further enrolment
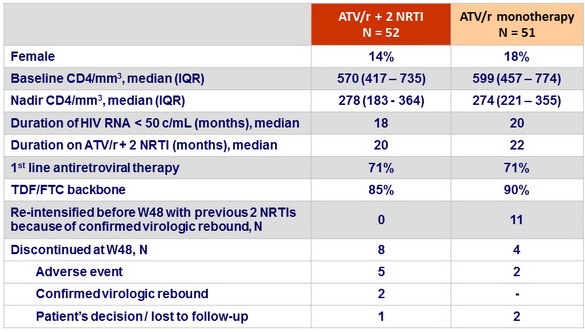
Baseline characteristics and disposition
Efficacy results
Safety, N (%)