Meynard JL. J Antimicrob Chemother. 2010 Nov;65(11):2436-44
Type of ARV Trial
Switch studies in virologically suppressed patients
» Switch to PI/r monotherapy
» LPV/r mono
Switch studies in virologically suppressed patients
» Switch to PI/r monotherapy
» LPV/r mono
Drugs
PI/r mono, LPV/r
PI/r mono, LPV/r
- In patients with virologic suppression, by per-protocol analysis, LPV/r monotherapy did not achieve non inferiority versus continuation of current antiretroviral regimen for maintaining HIV-1 RNA < 50 c/mL
- The incidence of virologic failure was low and successfully managed by treatment reintensification
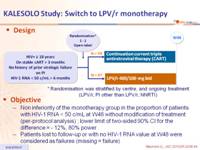
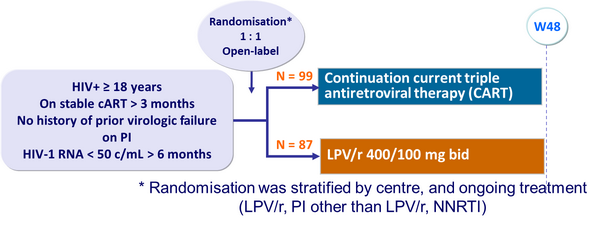
Design :
Objective :
- Non inferiority of the monotherapy group in the proportion of patients with HIV-1 RNA < 50 c/mL at W48 without modification of treatment (per-protocol analysis) ; lower limit of two-sided 90% CI for the difference = - 12%, 80% power
- Patients lost to follow-up or with no HIV-1 RNA value at W48 were considered as failures (missing = failure)
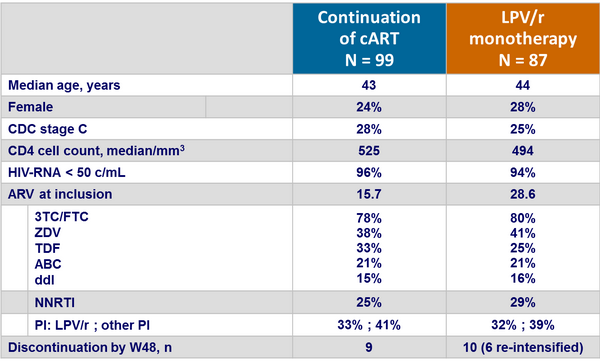
Baseline characteristics and patient disposition :
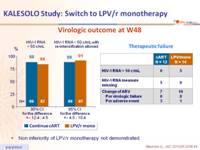
Virologic outcome at W48 :
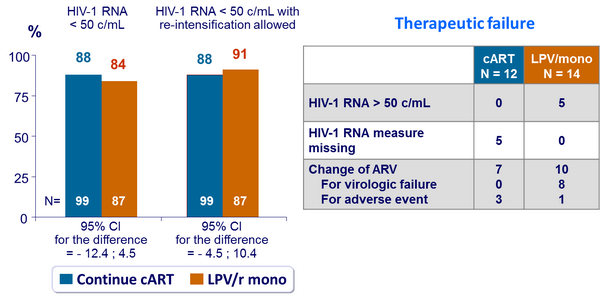
- Non inferiority of LPV/r monotherapy not demonstrated
Other outcomes :
- Multivariate analysis: treatment failure was not associated with adherence to therapy but was associated with older age at baseline
- Of samples with HIV-1 RNA > 50 c/mL, 20 genotypes from 14 patients (all in the LPV/r monotherapy group) could be analysed: 1 patient developed major PI resistance mutation, no patient developed mutations conferring resistance to LPV/r
- For lipids, fasting total cholesterol changes were significantly higher in the LPV/r monotherapy group (+ 0.42 mmol/L vs + 0.08 mmol/L ; p = 0.04)
- None of the 12 serious adverse events were drug-related
- Grade 3 to 4 laboratory abnormalities occurred in 3 patients in each group
- Diarrhoea was more frequent in the LPV/r monotherapy group (N = 34 vs N = 13 ; p < 0.001)